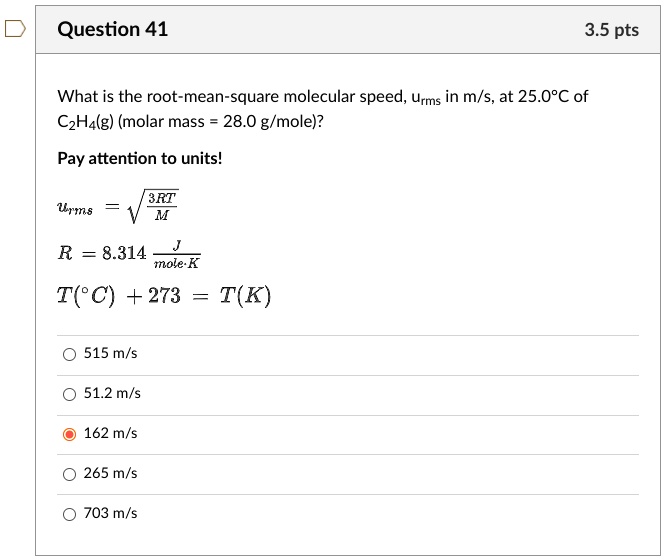
Initially, the root mean square (rms) velocity of N2 molecules at certain temperature is u . If this temperature is doubled and all the nitrogen molecules dissociate into nitrogen atoms, then the
Radial distributions of axial mean velocity (U) and rms velocity (Urms)... | Download Scientific Diagram

Turbulent channel flow: the root-mean-square fluctuation velocity U +... | Download Scientific Diagram

Root-mean-square velocity profiles normalized by the friction velocity... | Download Scientific Diagram

Mean (U) and root-mean-square (r.m.s.) of fluctuating (u rms , v rms )... | Download Scientific Diagram
the rms speed of Nitrogen molecule in a gas is u. if the temp. is doubled and the nitrogen molecule dissociate into nitrogen atoms the rms speed becomes

Which of the following relationships is valid for the root mean square speed (u("rms")) , average speed (u(av)), and the most probable speed (u ("mp"))?

Match of following (where U(rms)=root mean square speed U(av) =average speed U(mp) = most probable speed) {:(,"List-I",,"List-II"),("(a)",U(rms)//U (av),,(p)1.22),("(b)",U(av)//U(mp),,"(q)1.13"),((c),U(rms)//U (mp),,"(r)1.08):}

Identify the correct labels of A, B and C in the following graph from the options given below:Root mean square speed urms; most probable speed ump; Average speed uav

Identify the correct labels of A, B and C in the following graph from the options given below:Root mean square speed urms; most probable speed ump; Average speed uav
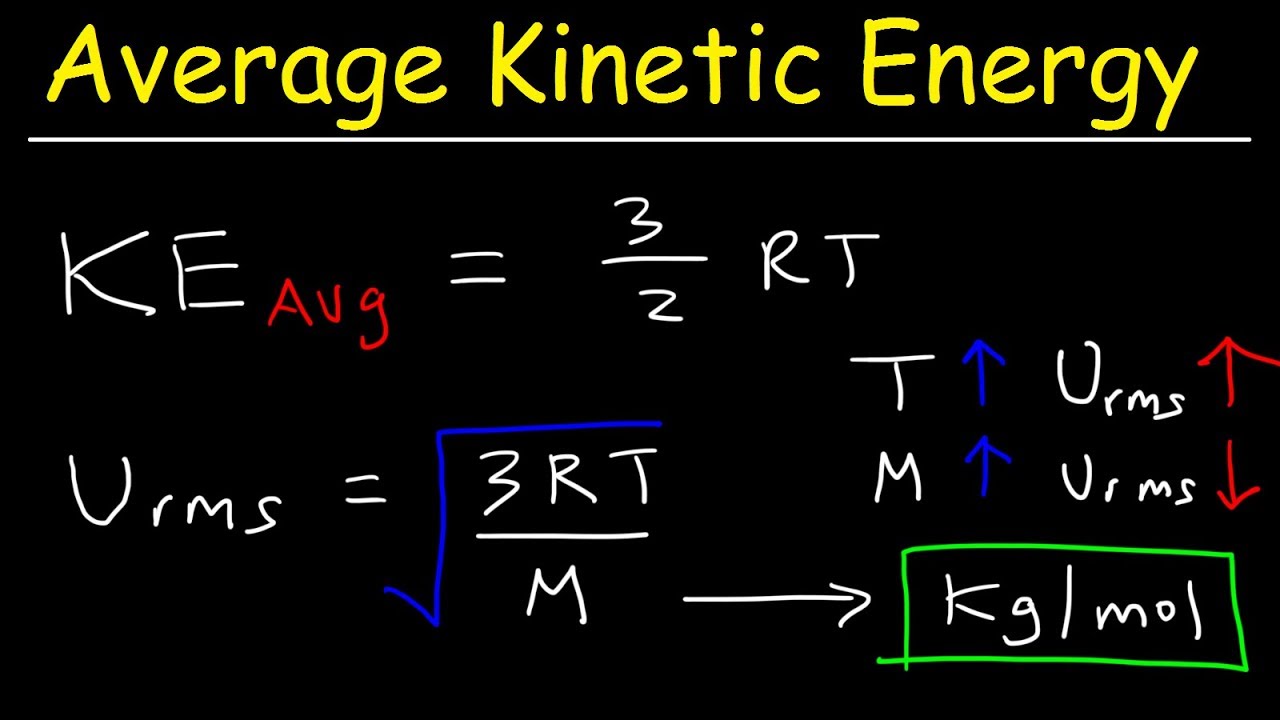
Average Kinetic Energy of a Gas and Root Mean Square Velocity Practice Problems - Chemistry Gas Laws - YouTube